What counts as a batch ID?
A batch ID is any identifier that ties your sample to a specific production run. The most common forms are a lot number printed on the vial label, or a manufacturing date printed on the kit box.
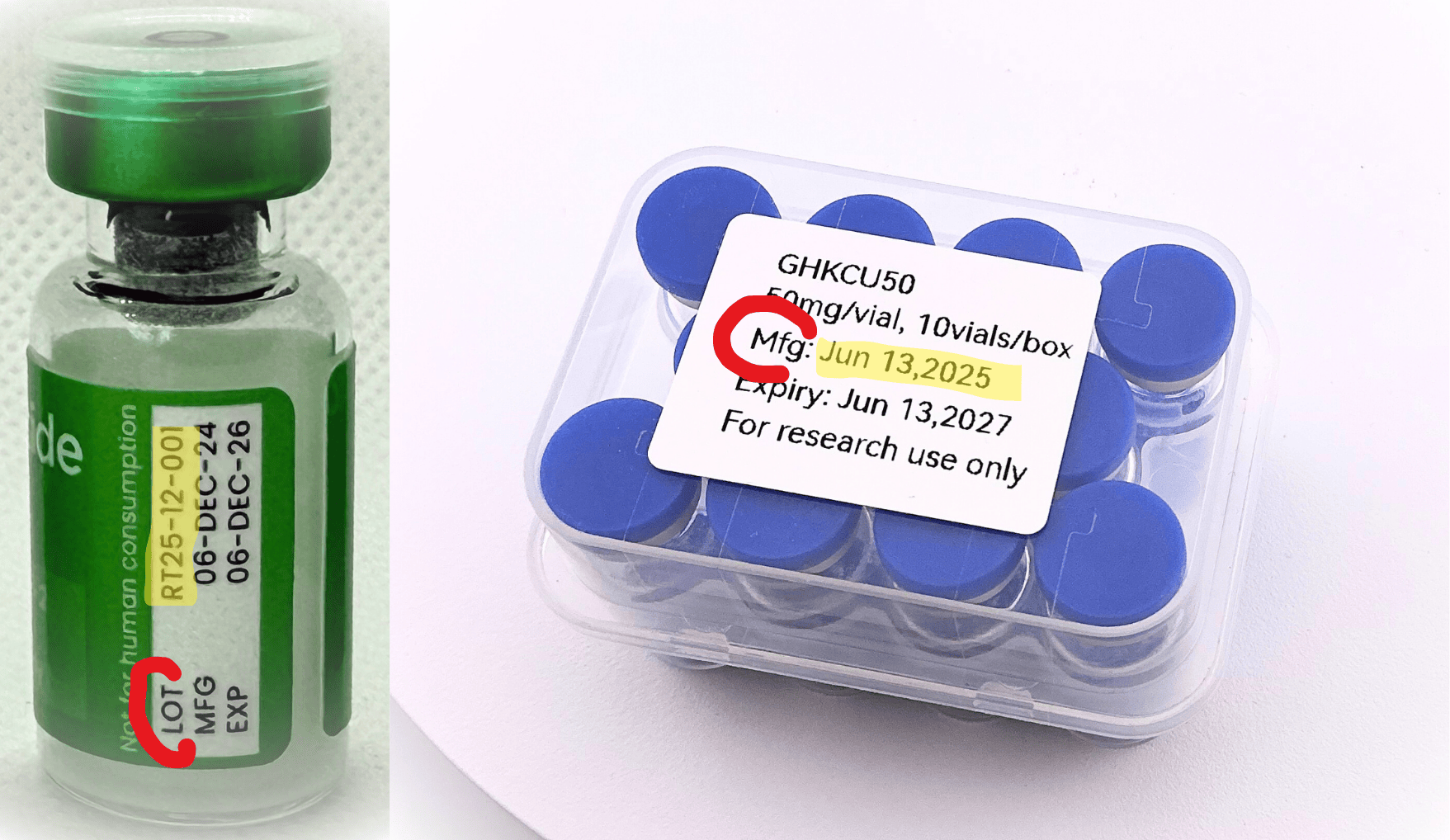
A batch ID is not a product name, a SKU, a catalog code, a CAS number, or an order number. If the only identifiers on your product are generic (for example, "BPC-157 5mg"), that is not a batch ID: it describes the product, not the production run.
If you're unsure whether something qualifies, include a clear photo of the full label and our review team will assess it. If your product genuinely does not carry a batch ID, you can still submit it for testing by selecting the "private" option or any paid add-on.
For vendors, we provide a basic guide to batch identifiers.
